
Valve-Regulated Lead-Acid (VRLA) batteries are the backbone of uninterruptible power systems (UPS), providing critical backup power in emergencies. However, understanding the factors leading to premature lead acid battery failure is essential for maintaining the integrity of these standby power systems. This article delves into the various elements that impact the longevity of VRLA batteries, highlighting the importance of proper battery care, usage, and maintenance to extend their service life.
Main Factors Affecting Battery Life
Service Life
Temperature
Overcharging
Undercharging
Thermal Runaway
Dehydration
Contamination
Catalysts
Service Life:
As defined by IEEE 1881, battery service life refers to the duration of effective operation under specific conditions, typically measured by the time or number of cycles until the battery's capacity drops to a certain percentage of its initial rated capacity.
In UPS (Uninterruptible Power Supplies) systems, batteries are generally maintained in a float charge state for the majority of their lifespan. In this context, a 'cycle' refers to the process where the battery is used (discharged) and then restored to full charge. The number of discharge and recharge cycles a lead-acid battery can undergo is finite. Each cycle slightly diminishes the battery's overall lifespan. Therefore, understanding the likely cycling demands based on the reliability of the local power grid is crucial during the battery selection process, as it significantly influences the risk of battery failure.
Temperature:
Temperature significantly affects how well and how long a battery works. When exploring how temperature impacts the failure of lead acid batteries, understanding the difference between ambient temperature (the temperature of the surrounding air) and internal temperature (the electrolyte's temperature) is essential. While the surrounding air or room temperature can affect the internal temperature, the change doesn't happen as quickly. For example, the room temperature might change a lot during the day, but the internal temperature may only see minor changes.
Battery manufacturers often recommend an optimal operating temperature, typically around 25 °C. It's worth noting that the figures generally refer to the internal temperature. The relationship between temperature and battery life is often quantified as a "half-life": for every 10 °C increase above the optimal 25 °C, the battery's life expectancy halves. The most significant risk with high temperatures is dehydration, where the battery's electrolyte evaporates. On the flip side, cooler temperatures might extend the battery's life but reduce its immediate energy availability.
Overcharging:
Overcharging refers to the process of applying too much charge to a battery, leading to potential damage. This issue might stem from human mistakes, like incorrect charger settings, or from a malfunctioning charger. In UPS systems, the charging voltage changes based on the charging phase. Typically, a battery will initially charge at a higher voltage (known as 'bulk charge') and then maintain at a lower voltage (known as 'float charge'). Excessive charging can significantly reduce a battery's lifespan and, in severe cases, cause thermal runaway. It's crucial for monitoring systems to identify and alert users to any instances of overcharging.
Undercharging:
Undercharging occurs when a battery receives less voltage than needed over an extended period, failing to maintain the necessary charge level. Persistently undercharging a battery results in decreased capacity and a shorter battery life. Both overcharging and undercharging are critical factors in battery failure. It should be carefully managed to ensure a correct voltage supply to maintain battery health and longevity.
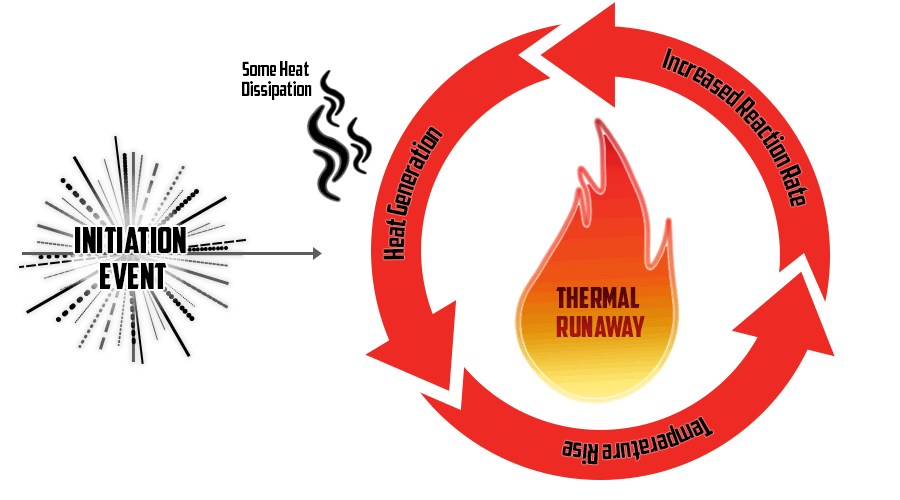
Thermal Runaway:
Thermal runaway represents a severe form of failure in lead acid batteries. When there's too much charging current due to an internal short or incorrect charging settings, heat increases resistance, which in turn generates more heat, spiraling up. Until the heat generated within a battery exceeds its capacity to cool down, thermal runaway occurs, causing the battery to dry up, ignite, or melt.
To combat this, several strategies exist to detect and prevent thermal runaway at its onset. One widely used method is temperature-compensated charging. As the temperature rises, the charging voltage is automatically reduced, and eventually, charging stops if necessary. This approach relies on temperature sensors placed on the battery cells to monitor heat levels. While some UPS systems and external chargers offer this feature, often, the crucial temperature sensors are optional.
Dehydration:
Both vented and VRLA batteries are susceptible to water loss. This dehydration can lead to diminished capacity and reduced battery life, emphasizing the need for regular maintenance checks. Vented batteries continuously lose water through evaporation. They are designed with visible indicators to check the electrolyte levels and easily refill water when needed.
Valve-Regulated Lead-Acid (VRLA) batteries contain much less electrolyte compared to vented types, and their casing is typically not transparent, making internal inspection challenging. Ideally, in VRLA batteries, the gasses produced from evaporation (hydrogen and oxygen) should recombine back into water within the unit. Yet, under conditions of excessive heat or pressure, the VRLA's safety valve might expel gas. While an infrequent release is normal and generally harmless, continuous gas expulsion is problematic. The loss of gases leads to irreversible dehydration of the battery, contributing to why VRLA batteries generally have a lifespan about half that of traditional flooded batteries (VLA).
Contamination:
Impurities within the battery electrolyte can severely impact performance. Regular checks and maintenance are vital, particularly for older or improperly maintained batteries, to avoid contamination-related issues. In Valve-Regulated Lead Acid (VRLA) batteries, the contamination of the electrolyte is an infrequent occurrence, often arising from manufacturing defects. However, contamination concerns are more prevalent in Vented Lead Acid (VLA) batteries, especially when water is added periodically to the electrolyte. Using impure water, like tap water instead of distilled water, can lead to contamination. Such contamination can significantly contribute to lead acid battery failure and should be diligently avoided to ensure battery performance.
Catalysts:
In VRLA batteries, catalysts can significantly enhance the recombination of hydrogen and oxygen, reducing the effects of drying out and thereby prolongs its lifespan. In some cases, catalysts can be installed after purchase as an additional accessory and may even help revitalize an older battery. However, it's important to proceed with caution; any field modifications carry risks like potential human error or contamination. Such alterations should only be undertaken by technicians with specific factory training to avoid failure to go into battery.
Conclusion
The premature failure of lead-acid batteries can be largely mitigated through proper understanding, monitoring, and maintenance. By recognizing the signs of potential issues like overcharging, undercharging, and thermal runaway, the life of VRLA batteries can be significantly extended. For those seeking further information and guidance, Dfun Tech provides comprehensive insights and solutions for maintaining the health and efficiency of lead-acid batteries. Understanding the intricate balance of physical and chemical factors that impact battery performance is crucial for anyone relying on these critical power backup systems.
Русский
Español
Deutsch
简体中文
ไทย
Français
English
Português
italiano
Bahasa Melayu
हिन्दी
Tiếng Việt
日本語
한국어
বাংলা
עברית
Latine
Nederlands
українська
தமிழ்
Polski
Türkçe
ພາສາລາວ
ភាសាខ្មែរ
ဗမာစာ
Filipino
Suomi
Ελληνικά
Svenska
Norsk
magyar
Română
Čeština
Монгол
қазақ
Српски
فارسی
Kiswahili
Slovenčina
Slovenščina
Հայերեն
Dansk
اردو
Shqip
Hrvatski
Afrikaans
Gaeilge
Eesti keel
Māori
සිංහල
नेपाली
Oʻzbekcha
latviešu
অসমীয়া
Aymara
Azərbaycan dili
Bamanankan
አማርኛ
Euskara
Беларуская мова
भोजपुरी
Bosanski
Български
Català
Cebuano
Corsu
ދިވެހި
डोग्रिड ने दी
Esperanto
Eʋegbe
Frysk
Galego
ქართული
guarani
ગુજરાતી
Kreyòl ayisyen
Hausa
ʻŌlelo Hawaiʻi
Hmoob
íslenska
Igbo
Ilocano
Basa Jawa
ಕನ್ನಡ
Kinyarwanda
गोंगेन हें नांव
Krio we dɛn kɔl Krio
Kurdî
Kurdî
Кыргызча
Lingala
Lietuvių
Oluganda
Lëtzebuergesch
Македонски
मैथिली
Malagasy
മലയാളം
Malti
मराठी
ꯃꯦꯇꯥꯏ (ꯃꯅꯤꯄꯨꯔꯤ) ꯴.
Mizo tawng
Chichewa
ଓଡ଼ିଆ
Afaan Oromoo
پښتو
ਪੰਜਾਬੀ
Runasimi
Gagana Samoa
संस्कृत
Gaelo Albannach
Sepeti
Sesotho
chiShona
سنڌي
Soomaali
Basa Sunda
Wikang Tagalog
Тоҷикӣ
Татарча
తెలుగు
ትግንያውያን
Xitsonga
Türkmençe
संस्कृत
ئۇيغۇرچە
Cymraeg
ייִדיש
Yorùbá
isiZulu


































